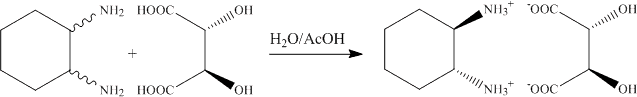
last updatedTuesday, January 12, 2016
Problems Meeting #2 (Condensed Key)
1.a. The addition of the trans-1,2-diaminocyclohexane to the L-(+)-tartaric acid solution leads to the precipitation of the (R, R)-diammonium salt. The (R,R)-enantiomer is isolated because of the matched pair geometry of the cation and anion that allows for strong interactions via six hydrogen bonds to three different anions (see X-ray structure discussed in lecture).
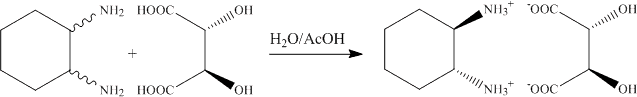
b. In the beginning of the experiment, the solution is acidic (concentrated aqueous solution of tartaric acid).
All amine functions are fully protonated due to the low pH-value. Thus, a small amount of the target compound will precipiate at this point that dissolves as the pH-value increases.
c. In order to precipitated the salt, both amine groups have to be fully protonated (pKa1= ~6.5). Thus, the pH-value has to be below pH=4, which is accomplished by adding glacial acetic acid (=100 % acetic acid).
d. Based on the information given in the reader and the lecture (w/v=1:2), the student should start with 8.4 mL =(4.2 g*2) of solvent for the recrystallization. This given ratio is lower than the one provided in the Hanson paper (w/v=1:10) because the student sample in Chem 30CL is still wet when the student starts the recrystallization.
e. Based on the quantities used in the reader, about 6.60 g of the tartrate salt should be isolated. The diamine (50.0 mmol =(6.0 mL*0.951 g/mL)/114.2 g/mol) is the limiting reagent here (tartaric acid: 3.75 g/150.09 g/mol= 25.0 mmol). One has to keep in mind that only 50 % of the total amount of amine are used here (=25.0 mmol). If the student isolated 3.00 g (=11.4 mmol= 3.00 g/264.2 g/mol)), the yield would be 45.4 % (=11.4 mmol/25.0 mmol *100 %) in terms of the (R,R)-enantiomer. Alternatively, he could also state that he isolated 22.7 % based on the total amount of diamine added. In either case, the yield falls below the range reported by the literature (60-70 % of the available enantiomer after recrystallization).
f. The tartrate salt is reacted with dilute sodium hydroxide solution to generate the free diamine, which is extracted into ethyl acetate. After drying the organic layer over potassium carbonate, a sample (~1.5 mL) of the organic layer is submitted for GC/MS analysis. Note that the solution is not concentrated or diluted.
2. a. Asymmetrically substituted sulfoxides are chiral because
they display a high inversion barrier. Thus, they are used as chiral
auxiliaries in asymmetric synthesis.
b. The stereoselectivity of a reaction fundamentally depends on the differences in activation energy (ΔG‡) of alternate pathways
of a reaction. The larger this difference is (i.e., induced by steric
hindrance), the more the rates differ resulting in a higher stereoselectivity. For a given difference in activation energies, the stereoselectivity
will increases with decreased temperature.
c. Oxazolidinones are usually obtained from amino acids, which can be isolated from the chiral pool.
d. Atropisomers are compounds that display a hindered rotation about a single bond. It is often found in ortho-substituted biphenyls or binaphthalenes (i.e.. 6,6'-dinitro-2,2'-diphenic acid, BINAP, BINOL, Knipholone).