last updated
NMR Simulations (has to be carried out in the SLC because the ACD program is not available elsewhere on campus!)
Learning Goals:
1. The purpose of this assignment is to
a. analyze the splitting patterns of various isomers and observe the changes in spectra when the structure changes
b. analyze the effect of concentration and nature of solvent on the
spectrum
c. analyze the effect of other NMR active nuclei in the
spectrum
2. Prediction of chromatographic results using simulated log P (=log Kow) values.
3. The classes will meet in YH 4340 (A, C and E) or YH 4346 (B,
D and F).
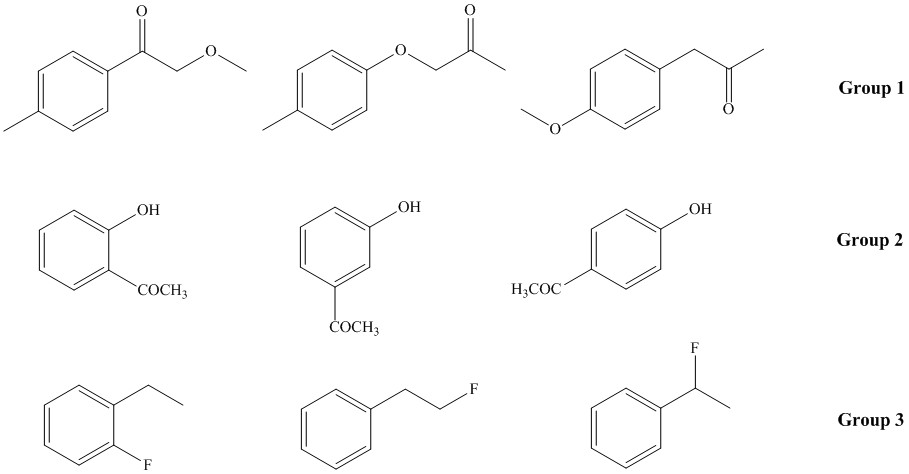
The assignment is mandatory and is done during the second half of the lab period in meeting 9 in the SLC. The assignment is due during your in-lab meeting in the meeting 10 in your lab notebook (credit: 15 points). Using Chemdraw and the ACDlabs 2015 LSM Software, simulate and analyze the the spectra for the following compounds:

General Comments
1. In order to draw these compounds, you will have to use ChemSketch, which is part of the ACD Software package (If the icon is not on the desktop, please check under ACDLabs 2015 LSM tab in programs). Try to make use of the existing templates available in the program i.e., benzene, etc. Otherwise, the drawing process is similar to the one in Spartan. However, you cannot draw the molecule in Spartan and then copy it over because the two programs use different data protocols!
2. To expand an area, click both mouse buttons. A crosshair appears on the screen and a second screen on the top. Define the left edge by moving the cursor to the left side and clicking the left mouse button. Then hold the left mouse button and drag the cursor to the right and click again. A dark gray field will move with the cursor to highlight the area to be expanded. Alternatively, you can also drag the markers on the top window to define the range.
3.It might be advisable to generated the expanded NMR spectra,
copy them to the report editor. From there they can be copied to a word file.
This will provide enough detail and also saves a lot of paper.
1H-NMR Spectra Simulations:
1. After you draw the molecules, highlight the molecule and go to "ACD/Labs-C+H NMR Predictor" to start the appropriate. Pick the "Calc HNMR" feature to simulate the H-NMR spectrum. Set the default frequency to 400 MHz in the beginning. (Options-Default Basic Frequency-500). The exchangable protons have to be activated in the "Show"-Menu. Go to "Option-Predict in Solvent" and check the "Chloroform-D" box only.
a. For group 1, analyze the peak locations and the splitting pattern for the aliphatic range. Rationalize the differences. How would you be able to determine the proper structure?
b. For group 2, analyze the splitting pattern of the aromatic range. Compare the obtained spectra with the literature spectra (=experimental data) that can be found on www.sdbs.com, www.sigmaaldrich.com, www.reaxys.com, etc. Do not use spectra here that have been simulated on a different website!
c. For 2-hydroxyacetophenone, go to "Option-Predict in Solvent", uncheck the "CDCl3-D" box and check the "Acetone-D6" box only. Click Recalculate. What changes in the H-NMR spectrum? Why? (Hint: Highlight the phenolic proton and right-click the mouse to get the chemical shift of this proton).
d. For group 3, analyze the splitting pattern of the aliphatic range and rationalize the locations. Make sure to activate the splitting with the fluorine atom (Options-Default Coupling-Check the H-F box).
2. Next, change the Default Basic Frequency to 60 MHz and recalculate the spectra for group 2. What changes in the spectrum? How can the changes be rationalized? Which of these frequencies (60 or 400 MHz) produces more useful data? Why?
3. Using the molecules of group 1, activate the Integration feature in the Show-Integral curve. Can you account for its shape and the steps observed?
4. For 2-hydroxyacetophenone determine the coupling constants (J3, J4 and J5) for the hydrogen atom adjacent to the the hydroxyl function by using Show-Table of Coupling Constants. What do these numbers mean in terms of the appearance of the signal for this proton in the H-NMR spectrum? (Make sure to activate the J5 coupling feature in Option-Default coupling).
13C-NMR Spectra Simulations (use 100 MHz frequency only):
1. Pick the "Calc CNMR" feature to simulate the C-NMR spectrum. Compare the different substitution patterns in the 13C-NMR spectrum of the molecules in group 2. (All generated spectra are proton-decoupled. Make sure to uncheck the Tools- Off-Resonance feature (How should signals look like at this point?). For the molecules in group 3, make sure to activate the C-F coupling by Options-Default Coupling-Check the C-F box).
2. For the molecules in group 1, check the Tools- Off-Resonance feature to deactivate the decoupling feature. How do the spectra change? Which conclusion can you draw from this type of spectra?
3. The program also allows you to do a "virtual lab". In the C-NMR module, go to the Show-Solvent lines item. A window pops up that allows you to change the solvent, its quantity and the sample weight. (Uncheck the Off-Resonance feature first!).
a. Using the Show-Solvent lines feature prepare a solution of 50 mg of 4-hydroxyacetophenone in 1.0 mL of (CD3)2SO (Don't forget to check the box on the top!). How does the spectrum look like? (Make sure to uncheck the Off-Resonance feature!).
b. Next, change the concentration to 1 mg of 4-hydroxyacetophenone and 1.0 mL CDCl3. Which changes do you observe in the spectrum? Rationalize this observation.
Chromatography Simulations:
1. Using the LogP simulator (Add-ons in Chem Sketch), determine the log P values for three molecules in group 2 as well as for 3-methoxyacetophenone.
a. Based on these values, determine the elution sequence of the
four compounds on a non-polar stationary phase (i.e., C18) with mobile phase of MeOH:H2O (60:40).
b. How would the situation change if a polar stationary
phase (i.e., SiO2) and and a mobile phase (i.e.,
EtOAc:hexane (20:80)) was used ?
Report:
1. Include all spectra (including their expansion if needed) simulated in this exercise (Hint: copy them into the report editor and from there into a word file (saves paper printing them out!).
2. Answer the questions above and rationalize the splitting patterns in the spectra.
3. The report for this project is due during meeting 10 (worth: 15 points). This assignment is mandatory!