Problem set #2 (Meeting 2)
Announcements:
1. The spectroscopy workshop will take place on Mondays from 5-6 pm in YH2200. The first workshop will be held on 1/22/2007.
ATTN: answers to the below questions are due at the start of meeting 2; these answers should be part of your pre-lab write-up. Show pertinent equations where appropriate.
1. Referring to the benzoin condensation, answer the following questions.
a. What is the function of the thiamine in the reaction and how is it obtained?
b. A student observes that the cap of the benzaldehyde bottle shows a lot of white crystals. Explain this observation. Which consequences does this have for the reaction?
c. A student starts off with 0.35 g of thiamine hydrochloride and 4 mL of 0.20 N sodium hydroxide solution. Afterwards, he adds 3.60 mL of benzaldehyde to the solution. After the work-up, he isolates 3.20 g of white crystals. Determine the yield for the reaction. Show your calculations.
2. Vacuum filtration
a. Draw a sketch for the setup used for the filtration of the benzoin and label all parts needed.
b. Why is it important to use filter paper in this step if a Hirsch funnel is used for filtration?
c. After the filtration, the crystals are rinsed with ice-cold water and ice-cold 95% ethanol. Why is this step performed and how does the student know that he rinsed sufficiently?
d. Why do the vacuum trap and the filter flask have to be clamped?
3. Referring to the elimination reaction, answer the following questions.
a. Why is it important to distill slowly during the first distillation?
b. What is collected during the first distillation?
c. After the distillation, saturated sodium chloride solution is used. Explain briefly why.
d. What is the function of the sodium sulfate used during the work-up? How does the student know that he added enough of it?
e. Which qualitative tests are performed to verify the presence of an C-C double bond? Show pertinent chemical equations.
4. Infrared Spectroscopy
a. How is the IR spectrum for the final product of the elimination obtained in the lab? Outline a brief procedure.
b. What are the most important changes in the IR spectrum going from cyclohexanol to cyclohexene?
5. a. Using PC Spartan 2002, perform the following calculations for the alkenes: terpinolene, limonene, a-terpinene, isoterpinolene. Make sure that all hydrogen atoms are present and there are also no extra ones either.
b. Select Calculations from the Setup menu.

The following window should appear. Select the options shown.
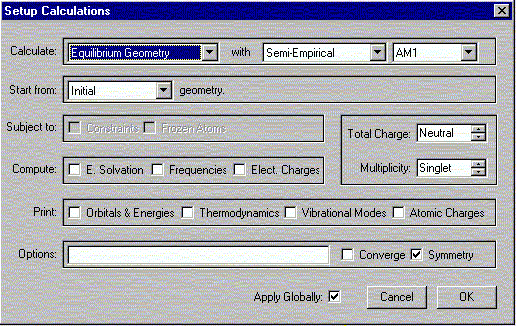
Verify that the Charge is Neutral and Multiplicity is Singlet. Click OK.
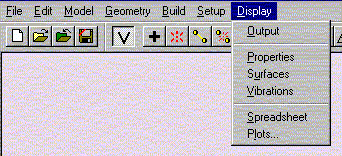
Select Submit from Setup menu.

Then click OK to exit dialog. Close the previous window.
Select Submit from Setup menu. When the calculation is completed you will be notified. It should not take more than one minute for the calculation to complete under normal circumstances. Make sure that you have only one window open at this point. The messages are usually displayed in the first window, and often not in the window you are working in at this point.
Under the Display menu, select Properties. Record the value for the Energy.
Analysis:
1. Based in the observed trends in trends in the heat of formation (=energy), which alkene is the most stable and which one is the least stable one? Can you rationalize these observations? Do the obtained data match with the expectation? If not, what is the conclusion?
2. Can you explain the product distribution observed for the elimination of water from a-terpinol (see reader page 20)?
Problems with calculations: Check here